One thing I have learned in my life is to question everything? Things are not always as they seem! For one to know or hold truth, one has to wade through a sea of lies. As a Live Blood Analyst and a Soil Microbiologist, I’ve had the pleasure and honor to see, study and discern the micro-world. This is both a science and an art! Over the years I have come to realize that when we observe the live blood on the slide, we are witnessing a living world that is communicating in more ways than what meets the eye. What is going on at this micro-level of existence?
Over the past few years, I’ve increased in concern for people’s anxiety and fear. It is probably the most rampant negative emotion that is having a negative effect on health. Yes, we have all gone through a pretty traumatic experience and paranoia is high!
I am not denying we live in a world full of toxins, chemicals and concern over dominion of control. There are threats and it is difficult to remove the “obstacle to cure”. I am not denying any of this. And, I don’t want those challenges or threats to be a burden, especially one that actually does more damage to a human’s health. If it is true we are under a silent and unseen attack, then instead of putting the fear out there, I’d prefer to offer hope and solutions. That is part of the mission of Living Ground and our work to be a “good little guy” education center.
“Knowing” the threats without having solutions or avenues to protect and provide an antidote, has serious consequences of emotional and physical health. The one true way to stay safe and healthy is to learn and follow nature and the miracle of nature. How do we do that? We observe nature! You do not have to be a scientist to do so!
To be honest, I am not seeing what others are under the microscope and if I do see strange things, I have other explanations. Some I have learned from the teachings of Antoine Béchamp, some from Dr Elaine Ingham and the rest is a questioning things and doing my own research.
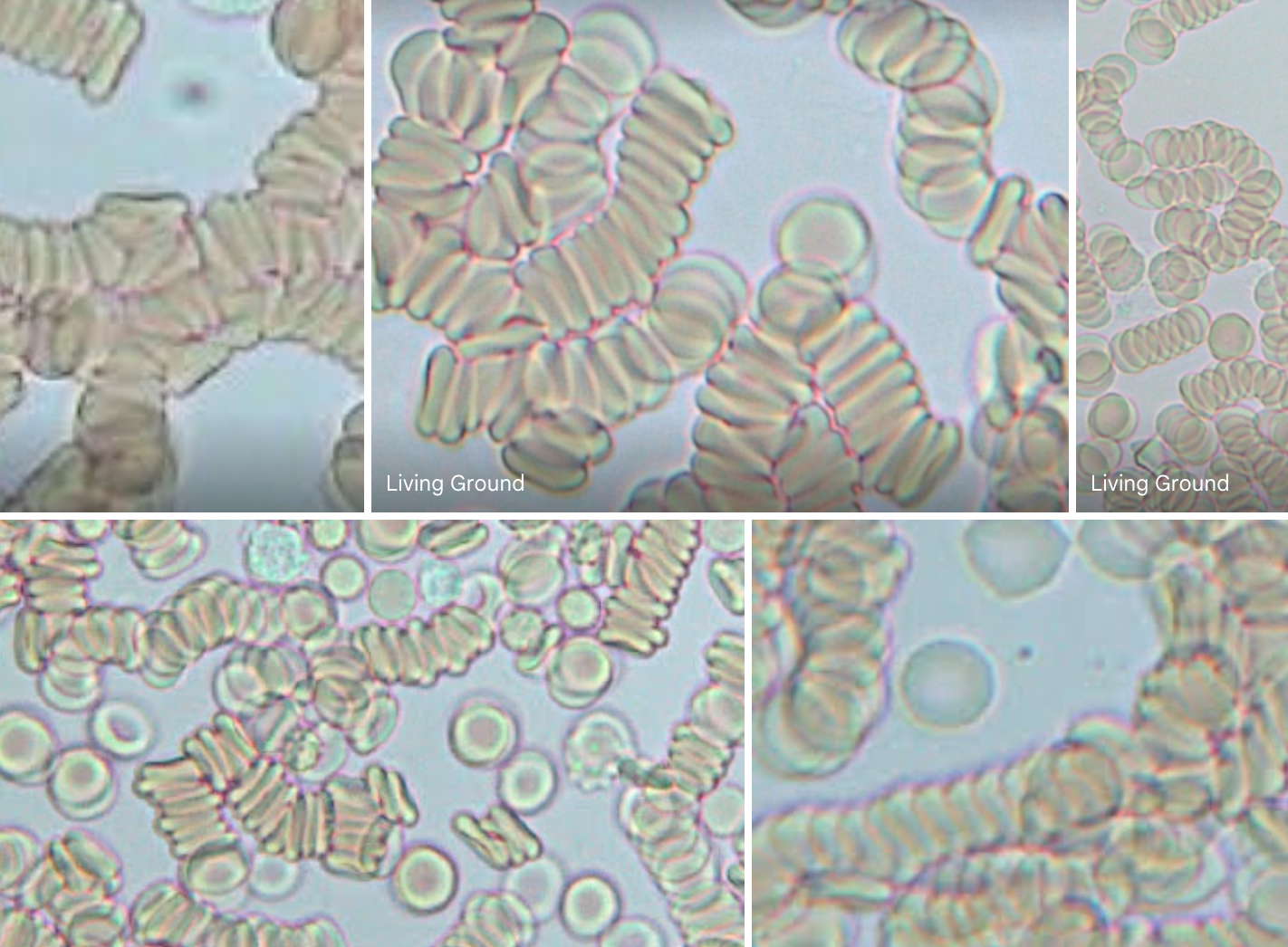
Many individuals are equipping themselves with microscopes. Substack posts and Rumble videos are showcasing what lies beneath the microscope’s lens, from geo-engineering aerosols in the atmosphere to innovative genetic drugs being introduced into our bodies. Among the presented findings is the term “microbial formations,” which is used to describe some of the phenomena witnessed in various bodily fluids, raising questions about their origins.
I do not follow germ theory that is the basis of most allopathic and natural (apparently alternative) approaches. What I mean is that many natural healers are following the germ theory. It is a war on pathogens. I practice and research pleomorphic and terrain theory. Although it is hard to see, the approaches are very different. Both these theories, Pleomorphic and Terrain recognize the dynamic relationship between microorganisms and the host’s internal environment. There is no war. While pleomorphic theory focuses on the adaptability of microorganisms, terrain theory places greater importance on maintaining a healthy internal terrain to prevent the proliferation of harmful microorganisms.
Both theories acknowledge the variability of microorganisms, particularly bacteria. Pleomorphism refers to the ability of certain microorganisms to change their shape, size, and characteristics in response to environmental conditions or the host’s internal terrain (or the environment on the slide). This variability can make it challenging to identify and classify specific microorganisms accurately.
Both theories suggest that microorganisms, including bacteria, fungi, and viruses (viruses are debated), are typically present in and on the human body. These microorganisms may become pathogenic or cause illness when the host’s terrain becomes compromised or imbalanced.
Considering these theories, if we discover a parasite and pathogen, the approach is different. Germ theory goes to war with “antibiotics” (natural or chemical) as a means to erradicate. Antibiotic means against life. What happens to the beneficials? Terrain theory is much like the new Soil Science that is discovering if you increase the beneficials (health), the pathogens leave! It is pro-life!
I share this article as I receive so many live blood videos showcasing these “entities”. My biggest concern is people’s reaction to these videos. Most, even if they are approach disease naturally are following germ theory. IMHO, this is dangerous! And, I am concerned for people’s emotional well being at this point because fear of germs and foreign objects is high!
The latest fear that is growing in the human mind is from the observation of nanoparticles in live blood. Let’s consider what a Nanoparticle is: they are typically defined as particles that have a size of between 1-100 nanometers (nm). Therefore, the micrometer (μm) size of nanoparticles is 0.001 to 0.1 micrometers. Can we even see this with a compound microscope?
And, then there are the Hydrogels. People are reporting seeing Hydrogels in live blood. Hydrogels are a type of polymeric material that can swell in water to form a gel-like structure. They are three-dimensional networks of hydrophilic polymers that can hold large amounts of water without dissolution. When viewed under a compound microscope with 1000x magnification, hydrogels may appear as clear or translucent structures with a soft, spongy texture. They can also exhibit a network-like pattern, depending on their chemical composition and preparation method. If a dye or fluorescent label is added to the hydrogel, it may appear colored or luminescent under the microscope.
No wonder people are afraid! Nanoparticles? Hydrogels? Is there a war happening at this microscopic world and do we need to be concerned about it?
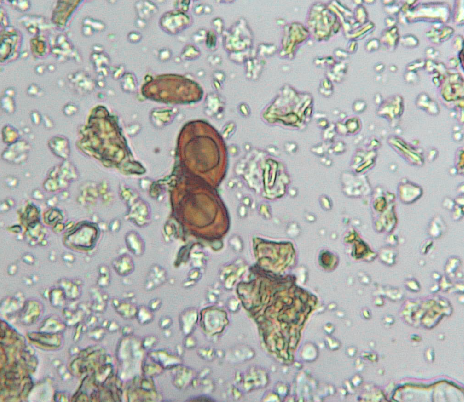
However, it’s essential to recognize that these formations are not entirely new and those who are “seeing” these forms in live blood might want to question a few things. I am! Yes, I do see crystals, symplasts of different shapes, sizes, colors. I do see pleomorphic objefcts. And, I’ve been forced to asked myself how are these “things” showing up in the blood on the slide?
So, let’s question some things….and use our common sense.
To start, most conventional compound microscopes have a maximum magnification of around 1000x. This is not sufficient to observe nanoparticles that are smaller than 50 – 100 nm. To observe nanoparticles at higher magnification, electron microscopy may be used. Electron microscopes use a beam of electrons instead of light and can achieve much higher magnifications, allowing scientists to study even objects as small as atoms. Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM) are two commonly used electron microscopy techniques to image nanoparticles.
The Hydrogels! Did you know that every cell within the human body is essentially a hydrogel, for cells inherently possess gel-like properties.
The videos and photos being shared with me, the structures observed vary in size, ranging from 150 microns in diameter to filamentous gel formations exceeding 1600 microns. Remember nanoparticles are 0.001 to 0.1
Let’s put this into perspective and let’s question things.
The typical size of a red blood cell, also known as an erythrocyte, is about 6-8 micrometers in diameter. To put it in simple terms, you could line up about 10 red blood cells next to each other, and they would be as wide as a single hair on your head. This blood exits the body via capillaries. Now, capilllaries are the smallest blood vessels in your body, even tinier than red blood cells. They are the tiniest blood vessels in the circulatory system. Their walls are composed of a single layer of endothelial cells, which is so thin that it allows for the exchange of oxygen, nutrients, and waste products between the blood and surrounding tissues. The diameter of a typical capillary ranges from about 5 to 10 micrometers (microns).
Did you catch that? Red blood cells are larger than capilliaries!
Because red blood cells are larger than capillaries, they must deform (do a yoga move) and squeeze through the narrow capillary passages to travel through the circulatory system, which is a critical part of their function in delivering oxygen and nutrients to tissues throughout the body.
So, here’s the interesting part. How does the blood get out? When a needle pricks your finger for a blood test, your body employs impressive mechanisms to help those tiny red blood cells leave the even tinier capillaries. Blood moves very slow in the capillaries. However, when poked, this acts as a gentle nudge that assists the blood, including the red blood cells, in exiting the capillary. The capillary’s elastic walls can stretch a little to create some space for the red blood cells to squeeze out due to the pressure from the poke. The walls of capillaries have nearly imperceptible gaps between their cells, like miniature doorways. Red blood cells are supple enough to change their structure to fit through these tiny gaps and exit the capillary. This is red blood cell yoga!
Additionally, when poked, your body dispatches signals, little messages to your blood cells, making the red blood cells more flexible and prepared to depart the capillary. Blood is still flowing even in the minute capillaries. As a result, when you’re stuck with a needle, the blood keeps moving, facilitating the movement of red blood cells out of the capillaries.
So, how do these other objects seen in live blood analysis appear? The nanoparticles that I am seeing in the videos and photos are MUCH LARGER than a red blood cell! How do they appear on the slide if it is physcially impossible for them to exit a tiny capillary?
There is anotherl point that needs to be considered through and through. The structures we are discussing in live blood may not present in your blood while it circulates in your body. Rather, they form later after a small sample of your blood is placed on a microscope slide.
During this microscopic examination of blood, we do and can see the formations. While the shape or structure may resemble crystals, symplasts, or other forms, it’s important to reiterate that they may not occur naturally in the bloodstream within your body and if they did, how did they exit the tiny capillaries? I am questioning this and how these formations arise when the blood sample is put on the microscope slide.
At this juncture, it is worth asking what constitutes a toxin? At its core, a toxin is anything for which the body lacks a sufficiently high inherent Zeta Potential (ZP) to disperse and eliminate when necessary.
Zeta Potential is a key property of colloidal particles, which are microscopic particles suspended in a liquid or gas medium. These particles can range in size from a few nanometers to several micrometers, and they can be found in various natural and man-made systems, such as soils, biological fluids, and industrial suspensions. Zeta potential describes the electrostatic charge distribution around a particle’s surface, which determines how it interacts with other particles and its surrounding fluid.
To understand zeta potential, it’s helpful to first consider the electric double layer that forms around a charged particle in a liquid medium. This double layer consists of an inner layer of ions that are strongly attracted to the particle’s surface, forming what is called the Stern layer, and an outer diffuse layer of ions that are attracted to the particle’s charge, but are not as tightly bound. This outer layer is called the Gouy-Chapman layer, and its thickness and charge density are influenced by the zeta potential of the particle.
Imagine your blood contains tiny particles, and some of them have either a positive or negative charge, like little magnets. Now, these charged particles create a kind of force field around them, and we call the strength of this force field the “zeta potential.” Basically it’s like a protective bubble around these particles. When the zeta potential is strong (either highly positive or negative), it’s like having powerful shields that keep these particles apart. They can’t clump together. When it is weak, they clump and stick together. The shields aren’t doing their job.
Is it possible this is how and why particles are forming in blood on the slide meaning they form due to the Zeta Potential? The zeta potential helps maintain the balance and proper behavior of the particles in your blood, preventing unusual formations like crystals and symplasts when it’s working correctly. This is one possible answer to my questions.
And, in the last year, I have been exploring Dr. Harvey Bigelson’s work and his perspective on blood communication and the formation of structures when blood is placed on a slide: Dr. Harvey Bigelson’s groundbreaking work in the field of holographic blood analysis is deepening my understanding of blood and its dynamic nature. His work proposes a unique perspective on blood, suggesting that it remains in communication with the individual even when placed on a microscope slide. According to Bigelson, blood is not merely an inert fluid but a living, communicative substance that responds to its environment.
“Living Blood”, In Bigelson’s view, is not a static entity but a living medium that maintains a continuous connection with the individual. When blood is placed on a slide, it is believed to retain its vitality and ability to respond to external stimuli. This perspective challenges the conventional notion that blood loses its inherent properties and communication abilities once outside the body.
Bigelson also suggests that blood, when exposed to various conditions or influences, can form specific structures as a means of communication. These structures may include crystals, symplasts, and other formations. According to his theory, these structures serve as a form of language or expression through which the blood conveys information about the individual’s health and well-being.
In other words, the blood samples on a slide are considered a form of biological communication. How cool is that? Am I looking at this deeper with my clients? Yes! Further, Bigelson contends that these structures are not random but purposeful responses to external factors. They may convey insights into the individual’s state of health, including information about toxins, imbalances, emotions, emotional and spiritual health or other health-related conditions.
So, in summary, yes, I question the formations people are reporting and that caused me to question what I do see! I question how these forms can exit the body from the capillaries (impossible) and I wonder if the formations are forming holographically to mirror a person’s fear? In other words, what you focus on (or afraid of) will appear on the slide!
This concept of “what you focus upon appears” suggests that our thoughts and attention play a powerful role in shaping our experiences and the world around us. If we focus our attention predominantly on positive things, we are more likely to experience positive outcomes and events, whereas if we focus on negative things, we may experience more negative outcomes.
Essentially, this concept operates on the idea that our thoughts and beliefs shape our reality, and that our minds have the ability to manifest our desires or fears into our physical experiences. This can be seen in various fields, including psychology, spirituality, and even physics. Perhaps it is true in live blood analysis too.
The concept of “what you focus upon appears” suggests that if a person is strongly focused on the idea of toxins or poisons being present in their body, there may be a greater likelihood of those structures appearing in the live blood sample due to the power of suggestion. However, this does not necessarily mean that toxins or other harmful substances are actually present in the body.
This is a personal perspective! I have done enough live blood and soil samples with the microscope that leaves me questioning more. I will continue the studying, researching, and exploring further. My questioning mind and the lack of observation of things presented by other live blood analysts has me wondering if my desire to help people clear fear influences what I see in the blood (and do not see)?
Regardless, I feel deeply and passionately that we need to find solutions and stop the fear!
Keep standing strong and expressing ultimate gratitude.












 We are Wild, the ones you cannot contain. We have no interest in being in your cultivated gardens. Rather, we eat the sun and cultivate each other, drawing on the life force energy of the dirt. Some years we ourish, creating abundance, and some years we almost fade away. These ever-returning cycles of life generating life are the way of the Wild.”
We are Wild, the ones you cannot contain. We have no interest in being in your cultivated gardens. Rather, we eat the sun and cultivate each other, drawing on the life force energy of the dirt. Some years we ourish, creating abundance, and some years we almost fade away. These ever-returning cycles of life generating life are the way of the Wild.” 















 Living Ground has been very busy working with clients to create their dream spaces. We are creating sustainable landscapes and vibrant ecosystem . Our recent project included a food forest, a medicinal and food spiral garden, a convenient kitchen herbal garden, activiating a pond and water system, and a water-efficient irrigation system from the nearby river.
Living Ground has been very busy working with clients to create their dream spaces. We are creating sustainable landscapes and vibrant ecosystem . Our recent project included a food forest, a medicinal and food spiral garden, a convenient kitchen herbal garden, activiating a pond and water system, and a water-efficient irrigation system from the nearby river.







 The Benefits of Our Soil Amendment
The Benefits of Our Soil Amendment
 Many flowers adapted with fire. They teach us that it is possible to survive and thrive even after a devastating experience. They remind us that fire is a natural and necessary part of life. The adaptation of certain flowering plants to thrive in fire-prone environments is nothing short of remarkable. Take, for instance Wild Hollyhock which I am attempting to grow in my garden. In the wilds, this plant used the heat of fire to trigger its seeds to germinate. These seeds are like memory-keepers, preserving the ancient wisdom of survival in extreme climates. What can we, as humans, learn from these eons of plant-gathered knowledge?
Many flowers adapted with fire. They teach us that it is possible to survive and thrive even after a devastating experience. They remind us that fire is a natural and necessary part of life. The adaptation of certain flowering plants to thrive in fire-prone environments is nothing short of remarkable. Take, for instance Wild Hollyhock which I am attempting to grow in my garden. In the wilds, this plant used the heat of fire to trigger its seeds to germinate. These seeds are like memory-keepers, preserving the ancient wisdom of survival in extreme climates. What can we, as humans, learn from these eons of plant-gathered knowledge?



























